Oral-History:Albert "Les" Babb
About Albert Babb
Albert “Les” Babb’s oral history is a fascinating life story of an engineer who crossed many disciplines of science and technology throughout his career. Born in Vancouver, British Columbia, in 1925, Babb’s father pushed him towards a career as a medical doctor, but Babb was drawn to chemical engineering. After graduating from the University of Illinois, Urbana-Champaign, he worked for Rayonier, Inc., but he was eventually drawn into an academic position with the University of Washington. Through his academic career, Babb was able to work in chemical engineering, but also in nuclear engineering (he was a cornerstone in the University’s fledgling nuclear engineering department). Eventually, he was drawn into biochemical engineering for medical advancements, a veritable full-circle back to his father’s original designs for him.
Although he has many engineering innovations to his credit, Albert “Les” Babb’s biochemical work on kidney dialysis machine advancement, sickle cell anemia, and pulmonary medicine research, have proved to be his greatest legacies. His gravitation towards the medical benefits of engineering innovations is facilitated by his sincere interest in the human side of technological advances and his goal to improve patient’s lives.
About the Interview
ALBERT "LES" BABB: An Interview Conducted by Frederik Nebeker, IEEE History Center, 6 December 2000
Interview #281 for the IEEE History Center, The Institute of Electrical and Electronics Engineers, Inc.
Copyright Statement
This manuscript is being made available for research purposes only. All literary rights in the manuscript, including the right to publish, are reserved to the IEEE History Center. No part of the manuscript may be quoted for publication without the written permission of the Director of IEEE History Center.
Request for permission to quote for publication should be addressed to the IEEE History Center Oral History Program, IEEE History Center, 445 Hoes Lane, Piscataway, NJ 08854 USA or ieee-history@ieee.org. It should include identification of the specific passages to be quoted, anticipated use of the passages, and identification of the user.
It is recommended that this oral history be cited as follows:
Albert Les Babb, an oral history conducted in 2000 by Frederik Nebeker, IEEE History Center, Piscataway, NJ, USA.
Interview
Interview: Les Babb
Interviewer: Frederik Nebeker
Date: December 6, 2000
Place: University Of Washington
Childhood, family, and education
Nebeker:
You were born on November 7, 1925 in Vancouver, British Columbia. Will you tell me a little about your family?
Babb:
During the first three years of my life in Vancouver I lived in the back of my father’s drugstore. That was located at Kitsilano Beach on English Bay overlooking Stanley Park and Vancouver, British Columbia, Canada.
Nebeker:
Was it part of Vancouver?
Babb:
Yes.
My earliest memory is riding a tricycle behind the counters in my father’s drugstore and helping myself to chocolates on display. I remember that quite vividly.
Nebeker:
I thought it interesting that you may have gotten the interests in chemistry and medicine from your father.
Babb:
Yes, I think so. My next memory is of my parents deciding to move south where there were good high schools and public schools. When I was three years old the family moved into what my father felt was the ultimate. It was a Spanish-style bungalow with a patio on 4170 Blenheim Street. My brother was born in 1928 and a sister arrived in 1931, so there were three of us. I went to a public school in the neighborhood, Lord Kitchener, and was active in the musical program and performing arts.
Nebeker:
Did you play an instrument?
Babb:
Yes, not very well, but I played a straight sax in the orchestra. I went to Point Grey Junior High School, south of where I was living. We commuted by bicycle. I went to high school near where we lived in 1939. I was active in athletics and music in high school. I learned to play the clarinet and was finally taught by the chief clarinetist of the symphony. I managed to keep up with everyone, but was not able to improvise. Therefore I knew that was not going to be my career.I was interested in science in high school was very much involved in athletics. I was captain of the Lord Byng High School junior rugby team when we won the regional finals. In those days everybody worked after school, and I had a paper route. I graduated from high school in 1943. By this time my father had made up his mind I should go to medical school. Consequently, I had taken four years of Latin in high school. He told me that I needed to learn that language.
University of British Columbia studies; employment
Nebeker:
Were you enthusiastic about medical school?
Babb:
I was and I wasn’t, but my father was always telling stories of his interactions with the doctors and himself as the person preparing the prescriptions. Physicians made house calls in those days. I was interested, but not to the point that I saw myself making a career of it. My first job after graduating from high school was at a metallurgical laboratory in Vancouver. They used to accept little sacks of ore prospectors brought in hoping that they were high in gold and silver content. I ground them and took samples. I got a nickel a sample. Cave & Company, the largest supply house of chemical glassware and chemicals, was next door. I got my hands on the freshman chemistry laboratory manual used at the University of British Columbia, so when I wasn’t preparing samples I was doing the experiments for freshman chemistry.
Nebeker:
Was this a summer job?
Babb:
Yes, this was right after my high school graduation. I did all the freshman chemistry experiments. There were professional chemists on site, so that helped.
Nebeker:
You had the equipment?
Babb:
I had the equipment and built a lab in my basement. Several windows got blown out in the process. My mother wasn’t too happy about that. Anyway, I did a lot of experiments at home and felt challenged by the idea of chemistry. My father was still saying I should really go to medical school, but I thought I should get an undergraduate degree before getting focused on what I should do. I entered the University of British Columbia in the autumn of 1943, and I immediately tried to enlist in the Reserve Officers Training Corps because we were at war with Japan and Germany. I flunked the physical exam because of my poor eyesight. The medical officer sensed my disappointment and said, “Why don’t you leave off wearing your glasses for 24 hours and come back tomorrow?” I did that, and of course I had memorized the big E. That gives you some idea of the motivation we all had at that time. He took me aside and said, “There is no way you can go on active duty. I’ll let you enlist in the ROTC because you’re so motivated, but I really think you should continue on with your education rather than enlisting. If you do enlist, you will be driving a truck in French Canada.”
Nebeker:
Like being sent to Siberia.
Babb:
Yes. To continue, I played varsity rugby for the freshman class and was also captain of the gymnastics team at the time. The summer that I spent at the metallurgical laboratory I worked out at the YMCA learning gymnastics. This all came to a halt. In the summer of 1944, after my freshman year of college. For example, we were sent by truck to Vancouver Island and north to a place called Comox that was adjacent to another town called Courtney. They were surrounded by huge cliffs. We spent two weeks training for amphibious assaults. As you can imagine, university students weren’t in very great shape. Our instructors were veterans of the Dieppe raid and had very little sympathy for college students. I survived that and then went to work for the B.C. Sugar Refinery. I thought I could get close to chemistry there. Along with a lot of other college students, I was a laborer trucking 100-pound sacks of sugar!
Nebeker:
This wasn’t your idea of chemistry.
Babb:
No, but I did get around to see the evaporators and equipment like that. Gradually I began to think more of the engineering of chemical processes rather than the chemistry of them. There was no medical school at the University of British Columbia at that time. At that time students in engineering all took the same courses for the first three years.
Nebeker:
Was this true for all types of engineers?
Babb:
Yes. Then they specialized in the fourth and fifth years. It was a five-year program, but it wasn’t as long as the academic year here because we were coming out of the Depression and everybody had to work for four months during the summer to earn our tuition and fees. I worked in a logging camp as a compass man and axe man in the summer of ’45. I worked out of a logging camp that was reached by a speeder. That was a little railroad car from Duncan, British Columbia. We arrived there with brand new caulk boots as they were called, new pants and new plaid shirts. We stayed overnight at the main bunkhouse before going out to the logging camp and were the butt of jokes from the old-time loggers, us college kids coming in with rosy cheeks and brand new gear. At the logging camp I couldn’t believe one could possibly sit before a more sumptuous table of food than was laid before us. It just staggered my mind. Breakfast? You name it, they had it. If the pancakes were cold, these loggers would dump the plates upside down on the table and call in the server they called a flunky.
Nebeker:
Had you chosen this summer job because you wanted the physical exercise or because that was what was available?
Babb:
It was what was available. Actually I loved being outdoors, and I learned how to take care of myself in the woods. We made our sack lunches at the logging camp. Loggers took us out to areas being surveyed for logging sites. It was hard work, but the crew was all college students and we became a very, very close-knit group. I was in the woods the summers of ’45 and ’46. To graduate one had to have a thesis on some kind of technical process.
Nebeker:
You specialized in chemical engineering?
Babb:
- Audio File
- MP3 Audio
(281 - babb - clip 1.mp3)
Yes, in my fourth and fifth year I specialized. I think there were twenty-five of us in that class. I went to work in a pulp mill at Ocean Falls after my fourth year. I don’t think I go back that far in my bio.
In the summer of ’47 I went to Ocean Falls and started working as a laborer in the pulp mill. Then I was assigned as a pipe fitter’s helper. The Mill was putting in a new black liquor recovery system for the Kraft mill, so I saw a lot of chemical engineering there. When something between 45- or 90-degree angles was needed I became sort of popular among the pipe fitters as “the kid with a slide rule.” I learned a lot about chemical processes there and the need for engineers to be more cognizant of what was needed when submitting blueprints. For instance we had to improvise our way around things like pipes specified to cross stairwells.
Graduate studies, U. of Illinois
Babb:
I was going to graduate a year later, and that’s when I started to wonder what I ought to do next. My favorite chemistry professor at the time, J. Allen Harris, used to come into my father’s drugstore quite often during the week just to browse through the magazine rack. My father had me apply to medical school at an eastern medical school and I received a tuition scholarship. Looking on a map, that school seemed like it could be in Siberia! On the other hand, Dr. Harris had published his dissertation at the University of Illinois in 1925. He had worked with Professor B. Smith Hopkins processing samples of monosite sands chemically by hand and decanting off the liquor, which they thought would contain element 61. Element 61 had not yet been assigned a name on the periodic table. They named it Illinium.They had competitors in Florence, where they named it Florentium. Then a group at Ohio State University started bombarding these sands with cyclotron radiation and found some spectral peaks they called cyclonium for element 61. Over the period of 1925-47 there were many different groups claiming they did it first.
In 1947 the documents from the Manhattan Project were just being declassified and, ironically, a group of chemists from MIT, on loan to the Atomic Bomb Project, had bombarded uranium in the pile they had at the Clinton Laboratories and in the fission products they found spectral lines that proved to them that element 61 was in the residue. That was the year before I was to graduate, and Dr. Harris took me aside and he said, “Look, I don’t think these guys are right. You would be doing me a great favor to go back to Illinois, to see my laboratory and to meet Professor Hopkins. I’m sure I can get you a fellowship.” And he did get me a fellowship to the chemical engineering department at the University of Illinois.
At the time, we were still coming out of the Depression even though there was more money available because of the war, and there was no way my family could support me. A tuition fellowship helped a lot, but I also needed room and board and books. It seemed incredibly far away and there were no commercial airplanes flying to that location at the time and it would be a long railroad trip. I told my father, “I think I’m going to go to the University of Illinois at Champagne-Urbana. As much as I’m interested in medicine, I think this is what I’ll do.” Dr. J. Allen Harris was very happy about that decision.
I graduated from UBC in 1948. I had a lot of interviews for jobs, but being from the west coast I was most interested in the one at Rayonier Incorporated in Shelton, WA, which is about 80 miles south of here. I joined them about July 1st, 1951. I had received my MS in ’49 and my Ph.D. in ’51.
Nebeker:
What was your dissertation work?
Babb:
My dissertation work was studying how molecules behaved when near a critical point, such as a saturated vapor condensing into a liquid. There was a theory that these molecules would agglomerate just before they would become part of the liquid phase. We used Raman spectra, shining lights of different wavelengths into a metal bomb and then looking at the spectrum of the reflected light. I did that for both my master’s and Ph.D.
Nebeker:
Who was your advisor?
Babb:
Dr. Harry G. Drickamer. He’s now retired, but still going strong and very active, and was a great influence on my career.
Nebeker:
Is he at Illinois?
Babb:
Yes. I had him out here five or six years ago to give a seminar. Two of my classmates are now quite famous. One is Walter Robb. He was from Penn State and was the director of the medical products division of General Electric and instrumental in marketing the CAT scan and the MRI scanners. He worked for President Jack Welch, who was also an alumnus of the University of Illinois chemical engineering department. He was about ten years behind me. Walt and I still keep in touch. He’s got a management and consulting firm now.
Rayonier, Inc.
Babb:
Anyway, back to Rayonier, Inc. I was very happy at Shelton, Washington. I liked the environment and I could go north on holidays to see my parents and brother and sister.
Nebeker:
What was your work at Rayonier?
Babb:
I was a research engineer and at the time I was working on a flow sheet for a new black liquor recovery system much like the one I had worked on in that pulp mill in British Columbia.
Nebeker:
Was this for a pulp mill?
Babb:
This was the research arm of Rayonier, Inc, and they had a pulp mill in Port Angeles, WA.
Nebeker:
Was that the intended use of this black liquor system?
Babb:
Yes. And being research-oriented, I saw that we needed a lot more data such as vapor pressures and other physical properties. I talked them into letting me do some studies, for example with sulfur dioxide in equilibrium with black liquor and not just water. I would come back to work at night. It wasn’t long before I was called into the Director’s office and said, “People are complaining about you working at night because they feel they ought to also.”
Nebeker:
You were setting a bad example.
Babb:
Yes. I had to quit that. Coming from a hothouse environment like the one at Illinois, the evenings were set up to come back to the lab.
High fidelity system design
Babb:
Now, to be busy in the evening, I took up high fidelity system design. I first purchased a mail order system. It was not stereo at that time of course.
Nebeker:
Right. Were you putting together a system yourself with an amplifier and speakers and so on?
Babb:
Yes. I designed a cabinet in which to put the tuner and record player. There was a company in town called Simpson Timber and some carpenters there built them for me. Then I obtained a great big bass woofer. This was when these things were not commonplace. And so that’s how I spent my evenings.
Nebeker:
That was the hi-fi movement.
Babb:
Yes. I renewed my interest in music and collected records. Television was not available at that time. Things were going along great.
University of Washington
Nebeker:
Were you married at that point?
Babb:
Yes. No children. I was getting regular salary increases, and the idea was that I would possibly in a year or eighteen months go to another facility, such as Jesup, Georgia. If I did well, then Rayonier would fund my way through Wharton Business School and get me headed on the management track. That pleased me a great deal. Coming through the Depression, having money to spend was not common. I had no interest in anything but having some “walking around money,” if you will and was very happy. That was 1951-52. Then in July of 1952 I got a letter from the chairman of this department.
Nebeker:
The chemical engineering department at the University of Washington.
Babb:
Yes. His name was Dr. R. Wells Moulton, and he asked me if I were interested in an academic position. Apparently they had written to the University of Illinois asking for faculty nominations. Since I was out here, it was an easy fix for them. It had never entered my mind that I would ever be in academia. I heard that they had huge workloads, low pay and requirements for research grants, so I hadn’t even thought about it. Anyway, I thought there was no harm checking it out, so I went up there one Saturday. At that time the department had only three members. Professor Moulton was chair. Another was Professor Joe McCarthy, who recently died. He was in the pulp and paper research business. The third was Professor Lennart Johannson. They wanted me to come back again and give a seminar. I went to the Director at Rayonier and he said, “We have to get permission from New York for you to talk about anything you’ve ever done, and not just what you’ve done here.”
I called Wells Moulton and said, “Excuse me. I can’t give you a seminar, but I can come up and show you the logbook in which I have accumulated a list of projects I would like to research if I ever had the opportunity.” I came up and showed it to the faculty, and by golly they sent me an offer. In retrospect, this was probably one of the toughest decisions I ever had to make. First, I had already gone against my father’s wish that I become a physician. Secondly, I was being tempted away from my goal of being in management. Finally, I didn’t know whether I could write research grants and didn’t know if I could be an effective teacher. Anyway, I decided I would accept the offer. I was young and didn’t have a family. That probably was the best decision I’ve ever made. Wells Moulton, who is about eight-five now, allowed me to do things I couldn’t do if I were a young faculty member nowadays.
Nebeker:
I take it that the university’s enrollment was increasing in those years and they needed new faculty.
Babb:
Yes. To digress, Dr. Moulton told me that the year before I came he was a human subject at the medical school, which had just begun graduating students. His brother-in-law was a pharmacologist and during the Korean War they were studying ways of preserving blood longer. He was part of an experiment where they would withdraw blood, do something to it and put it back. He got sick and his kidneys shut down. There was a young instructor at the Veterans Hospital named Belding Scribner, M.D. who was allegedly a nephrologist. That science didn’t exist in those days, but he was interested in electrolyte and fluid balance. He came over, and he had developed a bedside analytical kit for measuring sodium, potassium and so on. And he brought Moulton through this crisis. And as he told me about that, I just sort of filed it away, and I started the usual things that young faculty do. I was interested in diffusional processes at the time.
Nuclear engineering; reactor building and labs
Babb:
My academic progress was going along fine, and then Harold Wessman, the dean of engineering – also an Illinois graduate – called Wells and myself into his office. He was chairman of a joint committee of the American Society for Engineering Education (ASEE) and the AEC whose objective was to study how nuclear technology developed during World War II could be applied in a peaceful manner and what the university should be prepared to do to educate students in this area. He told us, “We want to establish a nuclear engineering educational program on the campus.” And he said, “Les, I want you to start getting ready for it.” I didn’t know a neutron from a fly on the wall. To make a long story short, I spent the summer of ’55 at Hanford working on what they call a Purex process for the separation of plutonium and uranium from fission products. That was actually was a decontamination process with respect to fission products and was something I was able to use very shortly thereafter when I met the medical people again.
Nebeker:
Was this separation procedure something with which you were familiar?
Babb:
No. I knew nothing about it. After the separation of plutonium, uranium and fission products, the plutonium was sent to Los Alamos for conversion into metal and then into atomic bombs.
Nebeker:
Was the separation procedure itself in the fluid state?
Babb:
Yes.
Nebeker:
Is it something like dialysis?
Babb:
The solvent extraction. Yes. They were pulsed solvent extraction columns. And when I came back then I was able to write a fairly large grant to the Atomic Energy Commission to study ways to design them to be more efficient. Then the next summer, in 1956, I went back with three other faculty members to the Argonne International School for Nuclear Science and Engineering at Argonne, Ill., and there I learned about scientific topics I hadn’t known about such as: neutron physics, reactor physics, and shielding design. They had a laboratory course for us where we could simulate separating plutonium and uranium. And a metallurgical lab where we made a few elements. It was very, very well run. There was one at Brookhaven too.
Nebeker:
The Atomic Energy Commission had the goal of promoting peaceful uses of atomic energy.
Babb:
Yes.
Nebeker:
I assume that at the time it was thought that a great deal of electric energy would be generated that way.
Babb:
Oh yes.
Nebeker:
Was that their main idea?
Babb:
Yes, that was the main idea. General Electric made a classic film called “A is for Atom.” It shows deserts being turned into wheat farms and airplanes, trains and buses propelled by nuclear reactors. It was euphoric at the time.
Nebeker:
Yes. Please continue.
Babb:
I went back to Argonne in 1957 after the dean decided the campus needed a nuclear reactor for teaching. At Argonne, we used a small teaching reactor called the Argonaut, which was available to us. We all learned how to bring the reactor critical and calibrate control rods. Back at the UW, we were controlling a mighty fission process, and it was impressive. We built an 18-ton graphite natural uranium pile across the street here in the basement of Bagley Hall. The uranium came from Hanford and the AEC arranged for us to borrow the fuel elements.
Nebeker:
Was that done within the chemical engineering department?
Babb:
No, it was within the college. A committee complied a program of courses and our first class began in the autumn of 1957.
- Audio File
- MP3 Audio
(281 - babb - clip 2.mp3)
It was a program that led to the MSE, which was already an established degree in the college. Therefore one didn’t have to fight the administration for an MS in nuclear engineering owing to the lack of such a department.Then one day the dean called me up said, “Les, we’ve got to find a site for this teaching reactor.” This was about 1958. We walked around the campus and found the best site, between mechanical and electrical and looking out over Lake Washington. It was on a gentle slope and the dean thought we could build into the bank and shield one end of the building. He said, “Why don’t you have one of your graduate students draw up a sketch?” I agreed, and he said, “We’ll make it out of cement blocks.” Therefore a graduate student and I put together a diagram of where the reactor would be located and laboratories and shops and things like that and took it back to him. He said, “That looks pretty good, but you’ve got too much glass.”
Those were the days when you could walk into the college president’s office and knock on his door, so we went to see President Schmitz. Having been a civil engineer, the dean figured he knew what the building cost would be and he said, “We’d like $80,000 to build this reactor building.” Schmitz put his hand on the dean’s knee and said, “Harold, there is no way you’re going put a cement block building on that beautiful site” and he included $350,000 in the budget for it. If you understand how things happen these days if you’re going to build a building then you need at least a ten year lead time!
Some of the first drawings were prepared by the Technical Artist Architect Group (TAAG). And they were artists! They had plans that looked like glass cages for animals in a zoo. If you look at a plan view of the campus, the civil engineering building and the mechanical engineering building, with Lake Washington being out here, the bounded space resembles a trapezoid. Therefore the artists thought we ought to have a building that’s trapezoidal in plan. I said, “Look you guys, there’s no way we’re going to build laboratories with a need for things like custom made lab benches. It just doesn’t make any sense.” They compromised by making a rectangular building with a trapezoid-shaped roof. These conferences occupied a lot of my time, but we were able to award a construction contract around 1959.
The reactor went critical in April of 1961, and we had a big dedication in June of that year.
Biomedical applications of nuclear reactor; cystic fibrosis screening
Babb:
One of the first tasks in what you might call the biomedical area had to do with a police investigation. The police chief from the Seattle Police Department showed up one day seeking our help. One guy was a suspect in the shooting of a neighbor in an adjacent trailer. The chief had a handful of bullets taken from the suspect’s trailer and a couple of slugs taken from the body of the deceased. He’d heard about neutron activation and asked, “Can you tell me if the ratio of antimony to tin in these bullets taken from the suspect’s trailer correspond to those taken from the decedent?”
Nebeker:
Didn’t they have the standard ballistics test looking at the scratches?
Babb:
They did, but wanted a chemical confirmation. We told him, “Chief, we’d love to do that, but there’s so much sodium and various salts in these bullets. Sodium activates very readily under neutron bombardment and the IR spectrum tends to mask the antimony and tin spectra, so we can’t do anything that would hold up in court.” However after the chief had gone a secretary sitting there, Marian Nelson, said, “My daughter has cystic fibrosis, and one of the manifestations of it is the abundance of sodium chloride in the sweat, fingernails and toenails. Maybe you should get together with the people at Children’s Hospital and see if you may have a way of screening newborns for the presence of cystic fibrosis so that they can get into early treatment.” Dr. Stanley Stamm came down and we talked about it. The result was that we set up a protocol whereby he advertised around the country and overseas that we were accepting toenails which would be irradiated with neutrons.
We accepted maybe a thousand samples from all over the world to irradiate. Here is a picture of a resident clipping the toenails of a baby at the University Hospital. There was a writer for Life magazine who lived in Belleview, just across Lake Washington, who did a story on this novel use of a nuclear reactor. That’s what got me associated with some clinical people.
A rheumatologist came over and was treating patients with gold salts. They wanted to do a material balance on them to see how much of the gold was being retained versus the amount excreted. We irradiated urine samples for him which was easy for us to do.
Nebeker:
Was that something else for which you could use the reactor?
Babb:
Yes. We could easily see the gold by irradiating the samples.
Nebeker:
Is there no simpler chemical test that would work?
Babb:
There probably is now, but it was very simple. It took less than an hour of irradiation.
Hemodialysis; central system for multi-patient dialysis
Babb:
To continue, early in 1963 Dr. Moulton called me into his office and said, “I just had a telephone call from Belding Scribner, M.D. He wanted to know if there’s a young faculty member here that could work with him in reducing the cost of hemodialysis.” I had never heard of this procedure. The reason Scribner called him was because he took care of Moulton when he was sick the year before I came. Suddenly Moulton said to me, “You’ve always been interested in medicine. Why don’t you go and see him?” Thus we made an arrangement. I went down to the University Hospital to a little room off the cafeteria. It was a daunting situation. There were about six physicians in white coats sitting there. They explained to me how they had just developed a way of permanently implanting plastic tubes in the artery and vein of patients, bringing them out through the skin and connecting them with a U-tube so that when they were not on dialysis the blood flowed at a high enough velocity to prevent clotting. Then they were clamped off and connected to the dialyzer with the artery going to the inlet port and the vein to the outlet port to start a dialysis episode.
Nebeker:
How long had that kind of extracorporeal dialysis been possible?
Babb:
It was first used during World War II. A physician named Wilem Kolff treated patients in Kampen in the south of Holland.
Nebeker:
Was it an advance in separation technique that made it possible?
Babb:
I’ll tell you how I looked at it. The Kampen had done this before, but they could not treat patients whose kidneys would never return to normal function. They were only able to keep the patency of the blood access to the cardiovascular system for a few weeks or a month at the most. If their function was not restored they would die.
After the war everyone was trying to develop permanent access. Scribner and his group had a four-layer dialyzer called a Skeggs-Leonards but it still hadn’t been able to provide permanent access to a patient’s cardiovascular system. Many patients came for treatment with this very crude system. If their kidney problem was chronic they would have to be sent home to die.
Nebeker:
Why didn’t they just put the catheters in every time?
Babb:
They ran out of access sites. You go up the arms and the legs and pretty soon you’re out of sites.
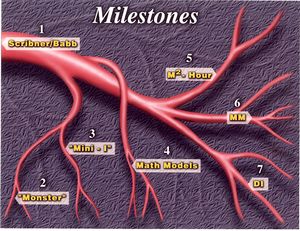
Recently, Scribner received a Lifetime Achievement Award from the Northwest Kidney Foundation and I received the Clyde Shields Award, as shown in this issue of Pledge, published by the Northwest Kidney Foundation and describes our collaborative work in detail.
To continue with our first meeting, a fellow drew a picture on the board of a hand showing how these tubes went in and came out on a plate, which patients wore on their arms. Scribner took a patient, Clyde Shields, and the mechanical engineer who had done the bending of the Teflon. They had spotted the Teflon on the insulation for electric wiring that was being installed in the hospital. A professor in dental materials said, “This Teflon is pretty slippery,” so it was tried and they were able to form it into eyedropper shapes at the end. In this way they could implant the tubes into an artery and vein and bring them all through the surface on this metal plate. And that was then the beginning of the possibility for chronic treatment of patients who would otherwise die.
Nebeker:
Was it the Teflon that was special about that connection?
Babb:
Yes. For the dialyzing fluid they started with a Sears Roebuck freezer. The dialyzer had two-layers made from polypropylene boards with grooves machined into them. Between them were two sheets of cellophane. The blood would come between them so that there were two parallel blood channels.
Nebeker:
I see.
Babb:
This Kiil dialyzer, named after the Norwegian inventor, was about 39” long, 13” wide and 6” deep and weighed about 60 pounds. The doctors then took me to the fifth floor of the Hospital and showed me the massive tank. When the Sears Roebuck freezers started rusting they thought about stainless steel tanks. There is a company in Seattle called Sweden Freezer. The president was an inventor, and I think he was the first one to make soft ice cream. Ironically, my father’s drug store in Vancouver had a soft ice cream machine made by Sweden Freezer. I worked at the drug store in the summer before going to college. I believe that my father’s was the first one in Western Canada to have the soft ice cream machine.
Little did I realize that one day I would meet Harvey Swenson, owner of Sweden Freezer. Anyway, Scribner called Harvey Swenson and said, “We need 400-liter or 100gallon stainless steel tanks and circulating pumps so we can pump dialysate through the outside of the parallel membranes.”
Nebeker:
Is this the dialysate that is on the outside?
Babb:
Yes, through the grooves. We have to be able to suck through the dialyzer so we can have a trans-membrane pressure to take water off the patients. If they’re not urinating to any great extent they are accumulating water, and that’s hard on the heart. Therefore, these pumps would suck the water mechanically. An osmotic pressure method with glucose was used before that, but that was very difficult to control.
Nebeker:
This was for a single patient.
Babb:
Yes, and right at the bedside. This tank had two pumps on it: one circulating the dialysate; the other pumped dialysate into the grooves outside the membrane. The patients’ blood was pumped by the heart between the layers of cellophane to remove toxins produced by the metabolism of protein, which were transferred to the dialysate.
Nebeker:
Was ordinary cellophane a suitable membrane there?
Babb:
Yes, believe it or not. In Holland Kolff used sausage casing because he was in the sausage capital of Europe!
Nebeker:
The accidents of history.
Babb:
He wound sausage casing around a rotating drum. He ingeniously figured out a way to connect a patient’s arterial side to a fitting that went into the sausage casing and came out the other end and back to the patient. And then this thing rotated in the dialysate bath. After the war it came out to Boston, but of course the people at the Mass General Hospital realized it was just too big. And it failed a lot.
I assimilated all this information, looked at the tanks in use, made some mental notes, and came back to my office in the nuclear reactor building. I had barely arrived when there was a phone call from Dr. Scribner, who had told me it cost about $20,000 per patient per year for two 12-hour dialysis sessions per week.
Nebeker:
Continue with the impact of this annual cost.
Babb:
Yes. By now people were hearing about this procedure and physicians were referring patients to the university, and we only had five of these tanks available. That’s when a committee was formed, the “Who Shall Live, Who Shall Die” committee of laymen. The committee consisted of one physician, a lawyer, a minister and a businessman. They established criteria for selection. Later, one person wrote that what we did was ethical, but someone else wrote that it was probably parallel to the Spanish Inquisition in terms of its lack of objectivity.
On that phone call to which I referred, I said to Scribner, “I take it your are interested in reducing the cost,” he said, “Yes, we’d like to get the cost down. Do you think you can help us?” And I said, “I need more information. I need to be walked through the whole procedure.” I took a stopwatch with me and it didn’t take long to figure out it was very labor intensive. There were just too many people involved. The tank had to be taken off the patient, pushed into an elevator, run down to the basement, emptied and sterilized. Then chemicals were weighed out, and the tank was filled, pushed back into the elevator and returned upstairs. It took three people to push the tank into the elevator. Plus, there were nurses hovering everywhere.
It dawned on me that what they were actually doing when they were trying to remove urea for example was decontaminating the patients’ blood with respect to these metabolites. That took me back to the Purex process we worked on at Hanford.
Nebeker:
Yes.
Babb:
I said okay, “This is a separations process.” I couldn’t get over watching these guys weigh out these salts. There must have been about five different salts. They’d weigh them out and dump them in. In the meantime, Scribner had contracted with a company in Minneapolis. They proposed that for each chemical they provide a little screw feeder with a hopper in which the chemicals are poured. The screw feeders were designed in the right proportion to give the right compositions with distilled water. From this idea we said to ourselves, “Why don’t we premix those chemicals in a concentrate and dilute it in the right proportion continuously. In this way we’ll remove the tank from each bedside and pipe the dialysate around the walls to maybe five stations where they can be connected to the dialyzers. The dialysate would flow through and then come back into a waste line. The mixing system and the other things would be outside the hospital room.”
We weren’t able to do that, unexpectedly, because the Fire Marshall would not let us put the machine outside in a corridor! We had to put it inside the room, which sort of ruined the elegance of it, if you can call it that, and it was dubbed “the monster!” Then all hell broke loose because manufacturers of commercial machinery besieged us, each wanting to be a supplier. I didn’t know much about patents, but having worked in industry I knew how important they were. I wrote up an invention disclosure and took it up to the business manager, Ernie Conrad, who was a good friend. In those days one could go from A to B without going through a mountain of bureaucracy. I said, “Ernie, I think we have something here that might generate millions if not billions of dollars. Do you think we ought to patent it?” And he sort of patted me on the head and said, “No. We’re an institution of higher education and in no way interested in commercialization. It’s university policy that if anyone asks for drawings or anything, we give them to them.”
Our friends at Sweden Freezer were close to our work and they were starting to put together their own version of our central proportioning system. At the time, the one who came out first with the best looking one was the Milton Roy Company of St. Petersburg, Florida. They flew me down as a consultant and had put together a very nice-looking console. I have a picture of that. They had a big wooden box sitting there. I asked, “What’s in the box?” and they said, “An anesthetized greyhound, as the patient.” It was because they have big vessels. I was there at the startup at the old VA Hospital in Coral Gables, Florida. From that beginning these central systems appeared all over the world.
Nebeker:
Were these multi-patient dialysis systems?
Babb:
- Audio File
- MP3 Audio
(281 - babb - clip 3.mp3)
Yes. Here we could handle five patients at once. Then Northwest Kidney Center was built in the nurses quarters of Swedish Hospital. They had an old time inventor as a patient. It would cost a fair amount of money to buy one Milton Roy unit, but this inventor built a proportioning system consisting of two screw feeders. One screw feeder was for the concentrate and the other for tempered water.
I had been very busy running the reactor and teaching nuclear engineering and chemical engineering. I had a lot of chemical engineering and nuclear engineering graduate students and my days were very long. We had just gotten this “monster” out in the sense that it was working and at half the cost, i.e. the annual patient cost was now about $10,000. A lot of the work we did after that was mathematical and we tried to model this process to see if it could be optimized. We suggested three 8-hour treatments per week rather than two 12-hour treatments were more optimal and convinced the medical staff to switch to this practice. By the beginning of 1964 I was feeling somewhat relieved that this was done.
Single-patient dialysis machine
Babb:
Then I got a call early one Saturday morning. Dr. Scribner was on the other end of the line, and his voice sounded heavy and depressed. He explained to me that a 15-year-old girl he’d seen many times had been turned down for treatment by the deciding committee I mentioned earlier. He told me she was going to die if she didn’t get treatment within four months, and the university had prohibited him from taking any more patients. He said, “Could you take this monster and miniaturize it?” At the time it had three pumps and was noisy, so the nurses called it “the monster” as I said before. He said, “It would be the same principle, diluting concentrate, but for one patient and portable so it could be taken to a patient’s home.” I said, “Aren’t we going to run into problems with the committee since they said no?” He said, “No. I propose that we make it a university research project. We’ll get funding for it outside the university from a foundation.” Then he added, “We’ve got four months to do this.”
I said, “I don’t know. I’ll have to talk to my engineers and technicians and see if they are willing to put in the time.” We were supposed to be doing what we were supposed to do for the educational programs. And I called them together and put the problem before them. And I said, “We’d have to make most of the stuff ourselves.” We couldn’t buy heat exchangers, for instance, that were this small, and the instruments weren’t readily available and we’d have to modify them. I said, “It really has to be almost automatic.” The patient could not be expected to do too much. We went for an automated system wherein at the push of a button the machine would pasteurize itself using hot water then. Then it needed to produce dialysate at the right temperature and composition and give an indication when one could hook up the blood lines. My staff said they’d like to do it, and I told Scribner we’d take a shot at it.
The nuclear reactor building was one of the Dean’s favorites, so he used to come over all the time to see what was happening. I knew if he came over and saw us working on this machine that I would probably be reprimanded. We had sliding doors or cabinets, where sliding one door over the other one would only see pliers and other tools, and if it were closed there would be a solid panel showing our drawings.
Nebeker:
I see, and you could cover them.
Babb:
We had “Dean watch.” Anyone up in the control room could see all over and warn us over the intercom that the Dean was coming. We’d slide the panels over and be doing what we were supposed to be doing by the time he arrived.
Nebeker:
This was a bootleg development.
Babb:
Yes. And I couldn’t have done it if I hadn’t had control of it. I could not do that here now. And at that time we happened to have probably the best cadre of electronics technicians on campus, and they were all under my supervision. We had a lot of ex-Navy people who had been in the nuclear propulsion program. We couldn’t have done it without them. If I had gone to this current chairman and said I wanted to do this, there would be no way. It was just the right time.
Nebeker:
How many people did you have working on this project?
Babb:
I would say about eight at the peak of it.
Nebeker:
Could you estimate the probability that the machine might fail and a patient die, for example?
Babb:
Yes, anything. We thought through many incidents fairly carefully except for one. Our water supply here was very pure, and we hadn’t looked beyond our own needs. We hadn’t thought about people in New Jersey and Downstate New York with hard water, or the water in California. We hadn’t given it any thought. Anyway, we got the single patient machine ready, and the young patient was trained at the University Hospital within the four-month period. We went to her home and directed the plumbers in what to do and fixed up her recreation room. I think there’s a picture of her in this scrapbook.
One of the factors that put pressure on me to build this machine was the fact that I knew her father, Harry, very well. He was an electronics representative and was in my office about every two weeks telling me about instruments we could use. My decision was heavily influenced by the fact that he was a friend whose daughter was dying. We got the plumbing put in and got the machine out there, and my techs went out there every week to deliver a tank of concentrate to her. And of course she was under the care of a physician.
Then I went up to Ernie Conrad again and said, “I think we ought to patent this.” He said, “No. You go back to your classroom and let us worry about the business.” People came from literally all over the world – Sweden, Denmark and so on – to photograph Mini I and take away drawings.
Nebeker:
There had not been any in-home single-patient dialysis units before?
Babb:
Except for these, no. There was one physician in England who took one of these Sweden Freezer tanks to a patient’s home and dialyzed her at home. But they had a nurse, doctor and technician there. However, our concept was for unattended dialysis overnight.
Nebeker:
This one allowed the patient to operate it herself or himself?
Babb:
Yes. It was for unattended overnight dialysis. Those interested in manufacturing these machines came from all over, so we had the drawings set up so they could photograph them. The first commercial machine that came out was based on our idea of automation. It was built by the same company in St. Petersburg, Florida. We ordered six of them and started training patients. We leased a floor of a motel near the campus for training. Our first patient was from Wisconsin and we trained the husband on the machine. Most of the others were from California. One of my dear friends in downstate New York ordered one of these and it plugged up on him because of the hard water. We hadn’t put in anything for water purification or water softening. I called the Milton Roy Company and said, “When you put these in, you’ve got to put in a water purification system,” and they did that, and they also came out with a simpler system. Then a group in Portland came out with a still simpler system that was more reliable and that was a real workhorse.Our original concept of a center, having dialysate made from one source, was good in principle but not in practice. This is because some patients need a little more or less potassium or magnesium. Therefore, since it wasn’t possible to do that dialysis centers started to go to the single patient systems. We had not envisioned that. Recently, as I went to several cities’ branches of the Northwest Kidney Center, I was amazed. The machines used the same proportioning principle and had better instruments and nice-looking heat exchangers manufactured especially for them. Still, they were a single patient unit. I guess those are what are most commonly used now all over the world. There are about 500,000 chronic dialysis patients in the world on these machines today.
Dialysis treatment article, 1997
Nebeker:
I wanted to ask about this 1997 article on dialysis treatment that was named a landmark article. What exactly was that article about?
Babb:
In addition to the mechanical design we were interested in what was happening to the patients when they were on the machine. We were looking to see if we could find ways to make life easier for the patient while reducing cost at the same time. We had a whole program modeling the patient artificial kidney system in parallel with this.
Nebeker:
Were you working on that with people in the medical school?
Babb:
Yes, Dr. Scribner and his Fellows. We did a lot of that. It would be better if I could show you slides of what we did.
Nebeker:
What was the subject of this article?
Babb:
The question was how to determine how many hours a week a patient should be dialyzed. We recognized early on that if a patient had any residual renal function that metabolites were being removed 24 hours a day. We saw that if we incorporated the residual renal function together with the dialyzer removal rate we could shorten the dialysis time in some patients. That led us to developing what we call the Dialysis Index for predicting the dialysis protocol. The renal function had to be specified, along with the body surface area, which we got from the patient’s weight, and also the efficiency of the dialyzer. Then we would come up with two times. One time was based on the small molecule removal requirement, for which we used creatinine as the marker, and the second was a middle molecule that had a molecular weight of about a thousand. Urea is about sixty. Creatinine is about 113. The longer of the two times is recommended. During those days I went into many dialysis centers, and they had a huge nomogram of the one that was in that article I showed you, which they used as a guide. The mathematical equations and simulation calculations were quite complex. Most doctors couldn’t be expected to understand the calculations so we put them together in this nomogram so that they could do the analysis with a ruler.
Nebeker:
Right.
Babb:
As you can see, that article was chosen as a landmark. We were continually working to understand the biochemical impact that dialysis was having on patients.
Nebeker:
That’s a very good story.
Engineering innovations in dialysis machines
Babb:
Dr. Scribner retired from research in the early 1980s. That meant there was little need for people like me. Things were pretty well established throughout the country.
Nebeker:
There must have been companies producing with their own engineers.
Babb:
Oh yes. We wrote that the best dialyzer would be short and should contain multiple parallel layers, rather than a long one with two layers. We showed that mathematically. Gambro Inc. of Sweden came out with one that was short and had about eight layers.
It was the Dow Chemical people that figured out a way of spinning hollow fibers. They came out with what we wanted – short, multiple channels. They would get membranes that were more permeable. This is a molded resin cylinder and contains about ten thousand fibers for one square meter of surface area.
Nebeker:
The blood is passing through the inside of the fibers?
Babb:
Yes. The dialysate is on the outside. This was a short many-channel dialyzer, which we had predicted mathematically. I think the ones that are most prominently used today are made in Japan.
Nebeker:
Do they use the same dialysate supply system with the hollow fiber dialyzer?
Babb:
Yes. That was a major technological breakthrough. As far as patients were concerned since they didn’t have to build a dialyzer like Caroline and her mother had to in their home with the first machine.
Nebeker:
Right.
Babb:
Now centers have ways of reusing dialyzers. They have a little machine that will flush them out and sterilize them. Patients go to centers but use single-patient machines and have their own dialyzers. The dialyzers are selling for about thirty dollars and they are getting typically three treatments from them. That makes them about ten bucks per treatment.
Nebeker:
That’s very efficient.
Babb:
That saves a lot of labor. It’s almost impossible to reuse these big two-layer Kiil dialyzers.
Sickle cell anemia experimentation
Nebeker:
Was there any connection between this and the sickle cell work?
Babb:
Yes. I can’t remember exactly what precipitated this, but some people from the sickle cell division of the National Institutes of Health came out to the center. They knew about our work in dialysis and so on and were sort of feeling us out. They told us about work that had been done at Rochester University and at the University of Kansas. They had taken blood from patients with sickle cell anemia. In sickle cell anemia the red cells look like sickles. Their experiments appeared to show that chemical agents could cause the cells to “desickle.”
Nebeker:
They collapse.
Babb:
Yes. They block capillaries. By accident, the University of Rochester had made a discovery. They thought urea might be a chemical that would bind to the end terminal valines of hemoglobin, and increases its oxygen-carrying capacity and to desickle the sickled cells so they wouldn’t be as rigid. They hadn’t used urea for some time and suddenly decided to do some ex vivo experiments. Incredibly, they found a change. It took them a while to figure out that urea decomposes to cyanate. They proved that it was the cyanate that was binding to the end terminal valine of hemoglobin rather than urea.
Then a hematologist at the University of Kansas took some sickle cell blood from a patient, put them in a plastic bag, poured in a cyanate solution and shook it up for a while. Then he analyzed the hemoglobin to see how much of the cyanate had reacted with the blood.
Nebeker:
Was he looking for the change in shape?
Babb:
Yes. And the NIH folks were out here telling us about this discovery for the first time, and they asked, “With your experience with extracorporeal technology might you be able to do this on a continuous basis?”
Nebeker:
What you needed to do was bring the cyanate in contact with the hemoglobin?
Babb:
Right. Then we’d have to dialyze off the unreacted cyanate so it wouldn’t go back into the patient’s body. We were starting to put together a protocol. First, they said they’d give us some money if we would study the kinetics of the reaction of cyanate with hemoglobin i.e., what order is the reaction and how long it would take to react for instance 40 percent of the cells, or something like that.
Nebeker:
Yes.
Babb:
I thought, “Well, it sounds like an interesting biochemical engineering experiment.” I got graduate students to study the kinetics, which we ultimately published. And then from the kinetics data we decided to do some extracorporeal studies. We got a 2-liter Erlenmeyer flask, some bank blood and started a continuous system of treating the blood with solutions of cyanate. We then determined the efficiency with which the cyanate reacted with hemoglobin. Then we studied the distribution of hemoglobin molecules that had been carbamylated as we called it, that had this reaction with the cyanate. We were looking to see what was the most probable number of sites we could carbamylate.The people from the NIH came out again and said, “How about building us a machine?” I said, “Okay.” We got a gift of an old kidney machine from our friend here in the Sweden Freezer company. To start, we actually built a microprocessor. We had a dialyzer to remove the unreacted cyanate, but we needed a detector that would shut the machine down if too much cyanate were about to come back into the patient. We had a concept and had to work out the details.
Then Beckton Dickenson Company wanted to fund some animal trials. I worked with Beckton and Dickenson, and we scoped out a machine that would include a kidney dialysis machine. It would provide dialysate and we could use hot dialysate to heat the blood in a bioreactor so the reaction would take place at a faster rate. Then we came up with a system that could detect unreacted cyanate.
Nebeker:
Was this so you could get a concentration measurement of the reaction?
Babb:
Yes. We put it all together and started experimenting on sheep. They don’t have sickle cell anemia, but we could find out if our kinetics were correct.
Nebeker:
Could you carbamylate their red blood cells?
Babb:
Yes. Ordinary blood cells can be carbamylated. We went through sheep trials and then built another machine using a kidney dialysis machine made and given to us by a company that used to be called Extracorporeal Medical Specialties. We designed the microprocessor to be in a separate cabinet that looked just like their kidney dialysis machine. This machine had little chemical pumps on the top. It also had the reaction vessel that we called a bio-reactor. A student who got an MS degree with me went to work for Extracorporeal. At the time we were asking them to wind a helix of plastic tubing with fishing line spacer.
Nebeker:
I see. To keep those apart.
Babb:
Yes. The blood would come in here, go around here and come out here. The dialysate came through here and would come out here. First we had to find out the limits of temperature that we could use without hemolyzing the blood. We ran experiments to learn the temperature limit and we had to find out the maximum concentration or the cyanate burden the patient could withstand with unreacted cyanate. We got this system working on sheep and we worked with hematologists who gave us many of the parameters we needed.
About this time the NIH asked about building three machines -- one for Kansas, one for Ohio State and one for us.
Nebeker:
What year was this?
Babb:
This was about 1981-84. An investor came out from Chicago and formed a company called Biomedix. He raised money from local investors. We did build these machines with the money he’d raised. We had given him the patent rights. The technicians from the other institutions came out here for training. I have a picture of one of our first patients here. It shows one vein being accessed with a needle.
Well, we finally got clinical trials under way. Unhappily, it turned out that for the effort and the cost we were not able to carbamylate or react enough of the hemoglobin with the sodium cyanate to cause a significant lessening in the number of crises they experienced. We would have to use a higher concentration of the drug with a higher risk to the patient. Therefore about 1985 or ’86 we decided that this extracorporeal technique probably was not going to be a viable treatment. That was a disappointment to all of us.
Nebeker:
Yes. And the company never actually produced?
Babb:
What happened was that the company then got into laser therapy of clogged veins in legs. The original investors came off pretty well in the end, but with another product.
Nebeker:
Has this idea of having the blood react at a higher temperature been taken up elsewhere?
Babb:
No. Now the emphasis is on a gene therapy technique.
Nebeker:
The gene therapy treatment rather than that kind of treatment. I was wondering if there is any other disease where this kind of treatment might be useful.
Babb:
There may be, Rik, but we haven’t found one yet.
Nebeker:
There is often a surprising transfer of technology when some other disease will be studied and someone will realize something like this can be used.
Babb:
That is so true.
Pulmonary medicine research
Nebeker:
How did you become involved in pulmonary medicine research?
Babb:
I’ve been modeling the interactions of how the lungs handle pollutant gases and how gases are distributed in the body. I’ve been doing that since 1985. I’ve graduated several Ph.D.s and written several papers about what could be called airway gas exchange.
Nebeker:
I see.
Babb:
The studies involve mathematical modeling and computer simulations.
Nebeker:
More science than engineering and trying to understand what’s happening with these pollutants in the lungs.
Babb:
Yes. Looking at how they are distributed and the paths through which they are removed from the body. For example alcohol and other ingested gasses. I’m going to retire from that in June. I retired from teaching in the Spring Quarter 2000.
I have one more Ph.D. candidate left, and he will graduate in June.
"The Genesis of the Square Meter Hour" publication
Nebeker:
Would you tell me a little about this citation classic, “The Genesis of the Square Meter Hour”?
Babb:
According to the square meter hour hypothesis, the amount of salt that is removed is dependent on the product of the area times the time. If the area is increased the time can be reduced. That was the theory.